Search results
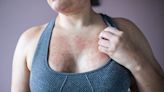
Supplements for Psoriasis: Which Ones Have Benefits or Risks?
Verywell Health via Yahoo News· 14 hours agoSupplements that may help improve or manage psoriasis symptoms include oral supplements like vitamin...
Refractory Psoriasis Can Be Overcome, Experts Say
MedPage Today· 6 days ago"While psoriasis/IBD/RA are all immune-mediated, they're inherently different disease states, and it's difficult to compare studies because of factors...
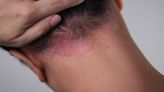
Skin Cancer and Rash: When Do Symptoms Suggest Cancer?
Verywell Health via Yahoo News· 17 hours agoIt has a variety of appearances and may mimic psoriasis or eczema, but doesn’t cause a widespread...
Are your prescription drugs affordable? Colorado board wants feedback on these 2
KDVR Denver via Yahoo News· 7 hours agoDENVER (KDVR) — A Colorado board continues reviewing the cost of prescription drugs, and public...
8 emerging uses for medical cannabis
Stacker via AOL· 16 hours agoIn 2018, the Food and Drug Administration approved a CBD medication to treat seizures associated with a severe form of epilepsy. VGstockstudio //...
FDA Approves Second Ustekinumab Biosimilar
Medscape· 1 day agoThe biosimilar is expected to be marketed in the United States on or after February 21, 2025.
What is perioral dermatitis? Treatment, symptoms and causes
Cosmo via Yahoo News· 16 hours agoCosmopolitan UK answers your most searched questions on Hailey Bieber's skin condition. What is...
Why do we lose so much hair?
Live Science via Yahoo News· 23 hours agoBoth men and women can have either of these hair-loss patterns, depending on the levels of certain hormones. Scalp health can also affect how much hair...
Small Business - The Brewton Standard | The Brewton Standard
The Brewton Standard· 2 days agoNEW YORK, April 16, 2024 (GLOBE NEWSWIRE) -- Attorney Advertising -- Bronstein, Gewirtz & Grossman, LLC, a nationally recognized law firm, notifies investors that a class action lawsuit has been filed against Ventyx Biosciences, Inc. (“Ventyx
Alvotech and Teva Announce U.S. FDA Approval of SELARSDI™ (ustekinumab-aekn), biosimilar to Stelara®...
Morningstar· 1 day agoFood and Drug Administration (FDA) has approved SELARSDI (ustekinumab-aekn) injection for subcutaneous use, as a biosimilar to Stelara®, for the treatment of moderate to severe plaque